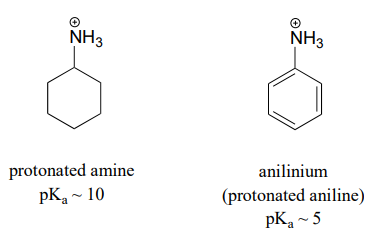
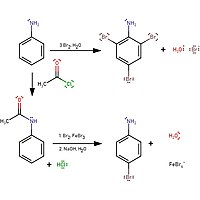
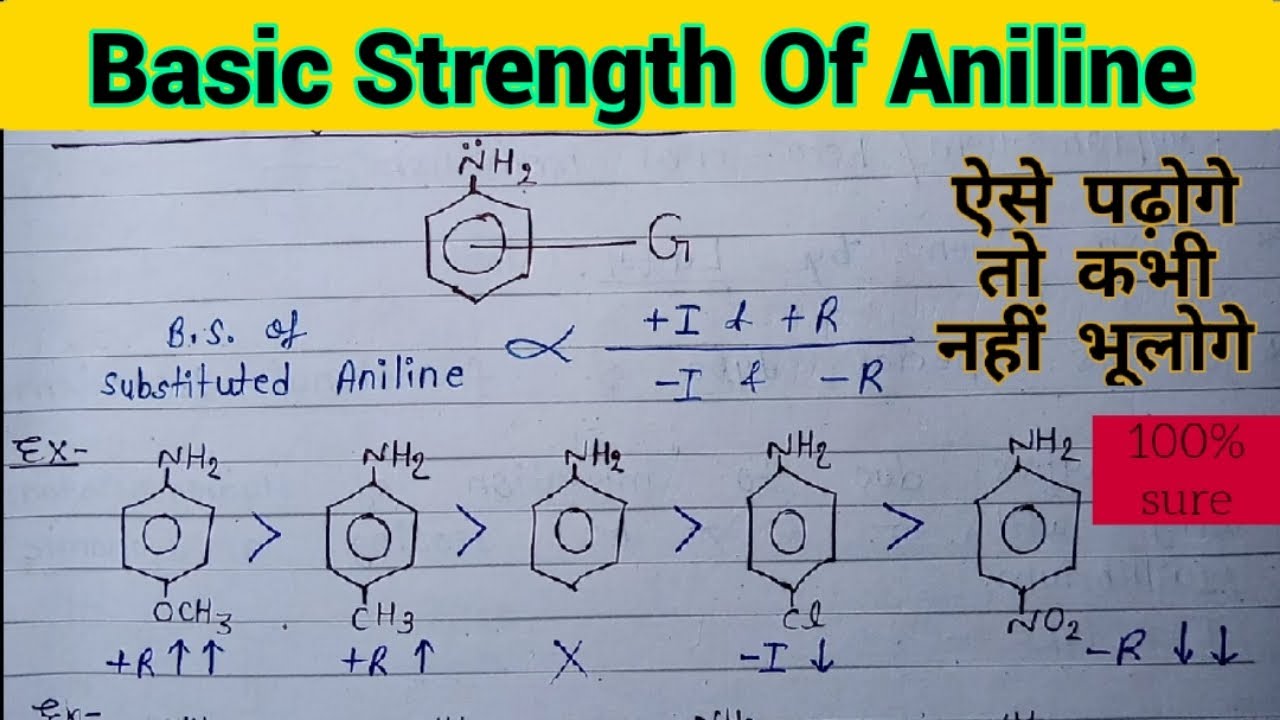
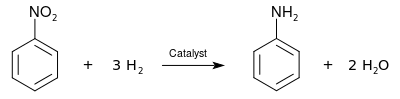
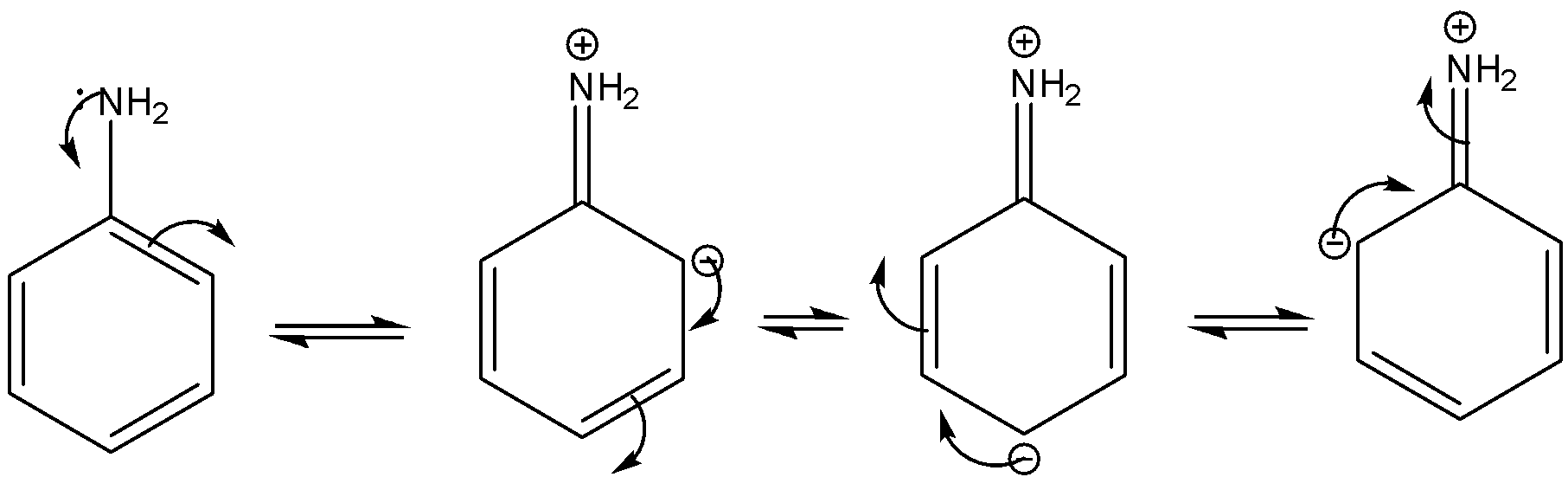
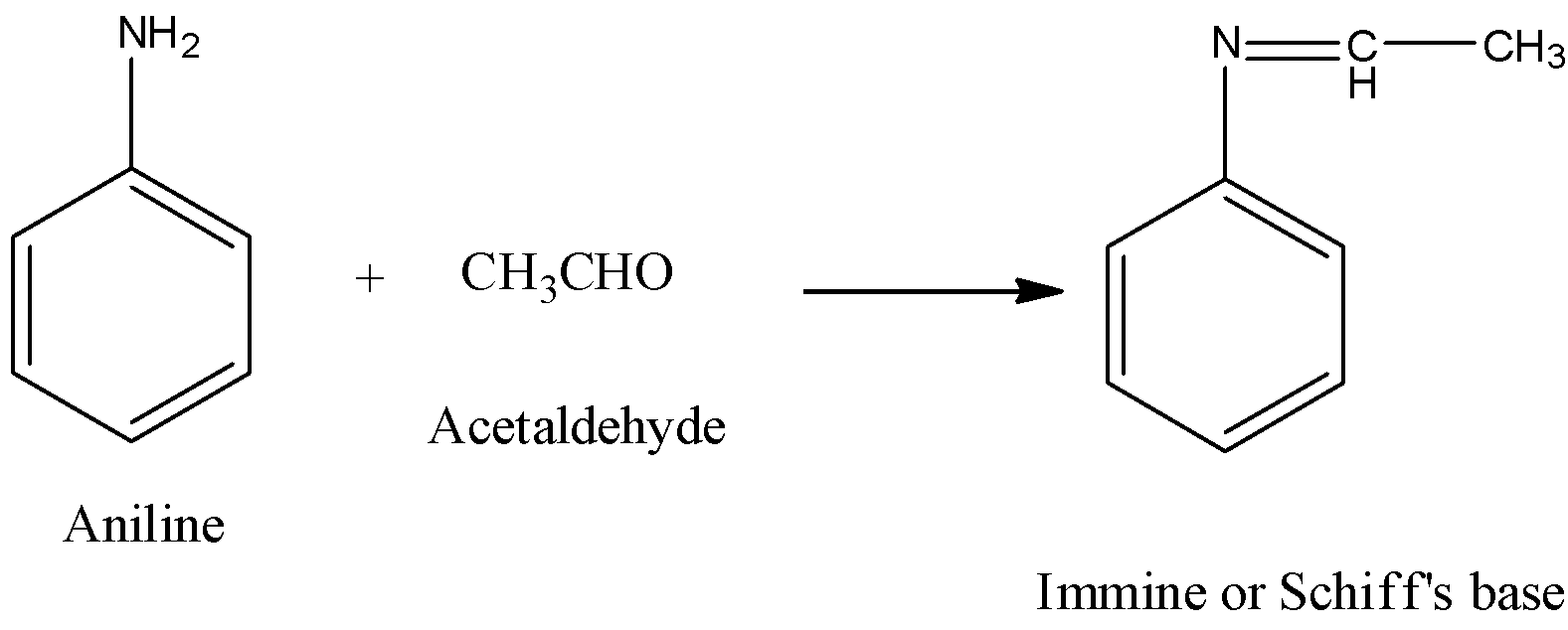
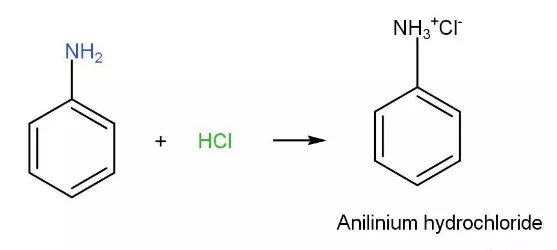
organic chemistry - Which is the strongest base among the given anilines? - Chemistry Stack Exchange

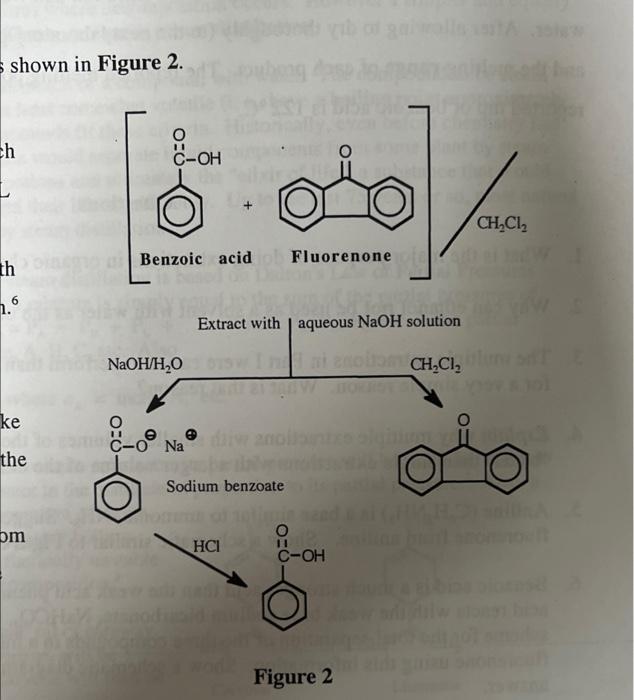
Propose a method to separate the following three compounds: benzoic acid, aniline and naphthalene. How would this separation look in the form of a schematic diagram? | Homework.Study.com
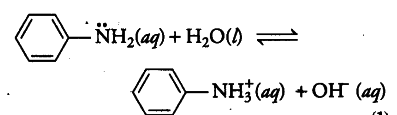
Write the equilibrium equation for the acid-base reaction - CBSE Class 12 Chemistry - Learn CBSE Forum
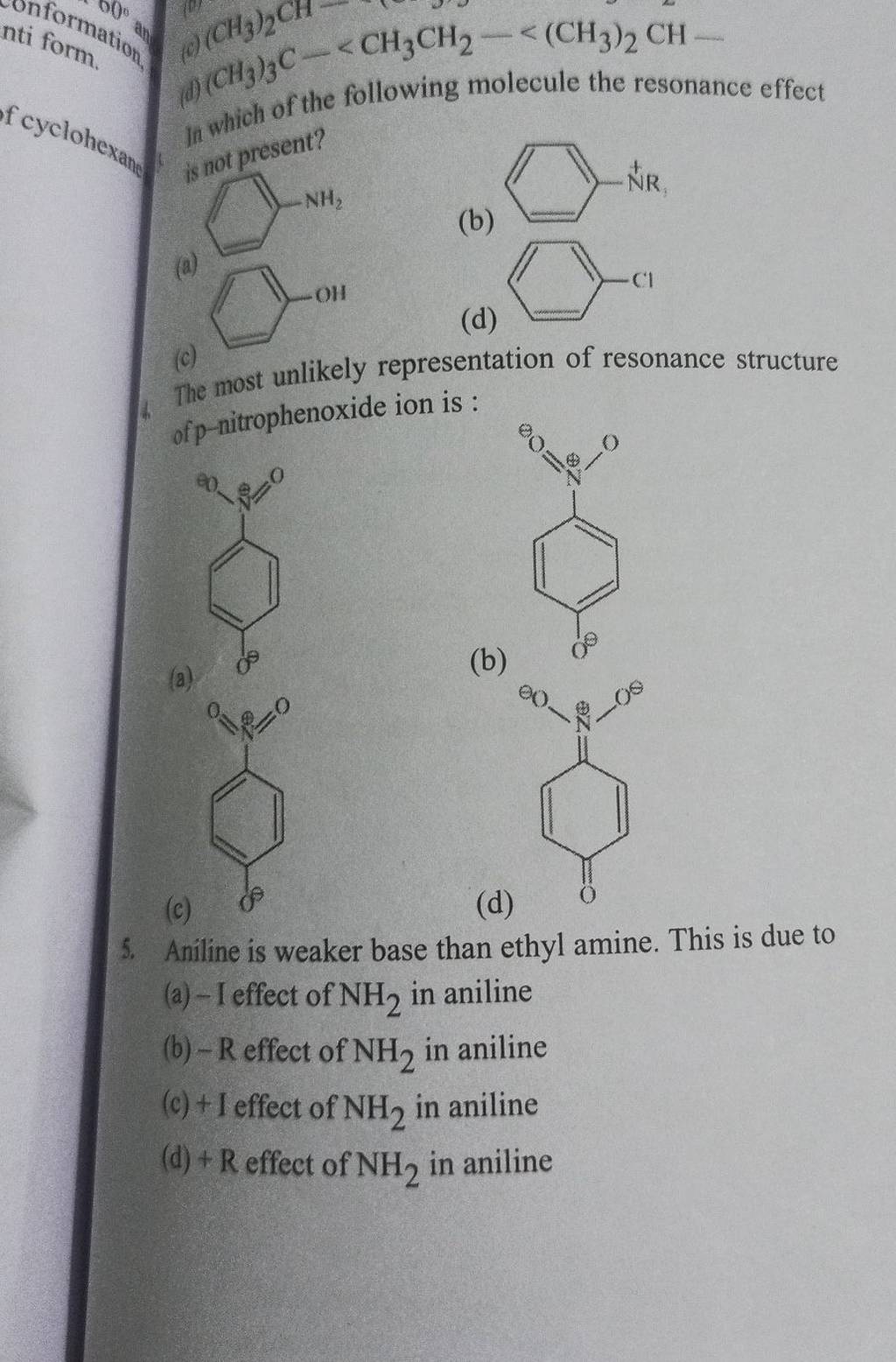
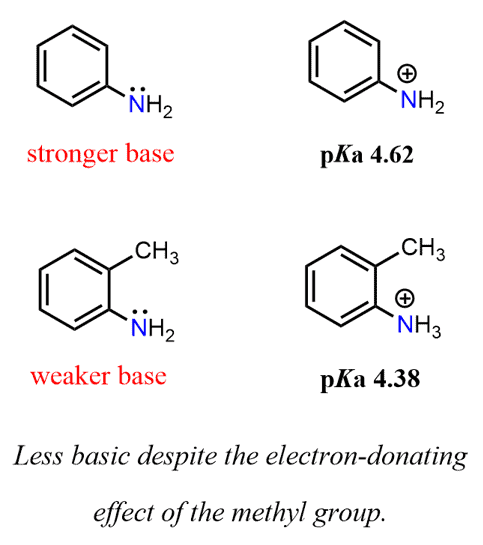
Arrange the following as directed: (a) Increasing order of basic strength: Aniline, p – nitroaniline and p – toluidine. (b) Decreasing order of basic strength in gas phase: C2H5NH2, (C2H5)2NH, (C2H5)3N and