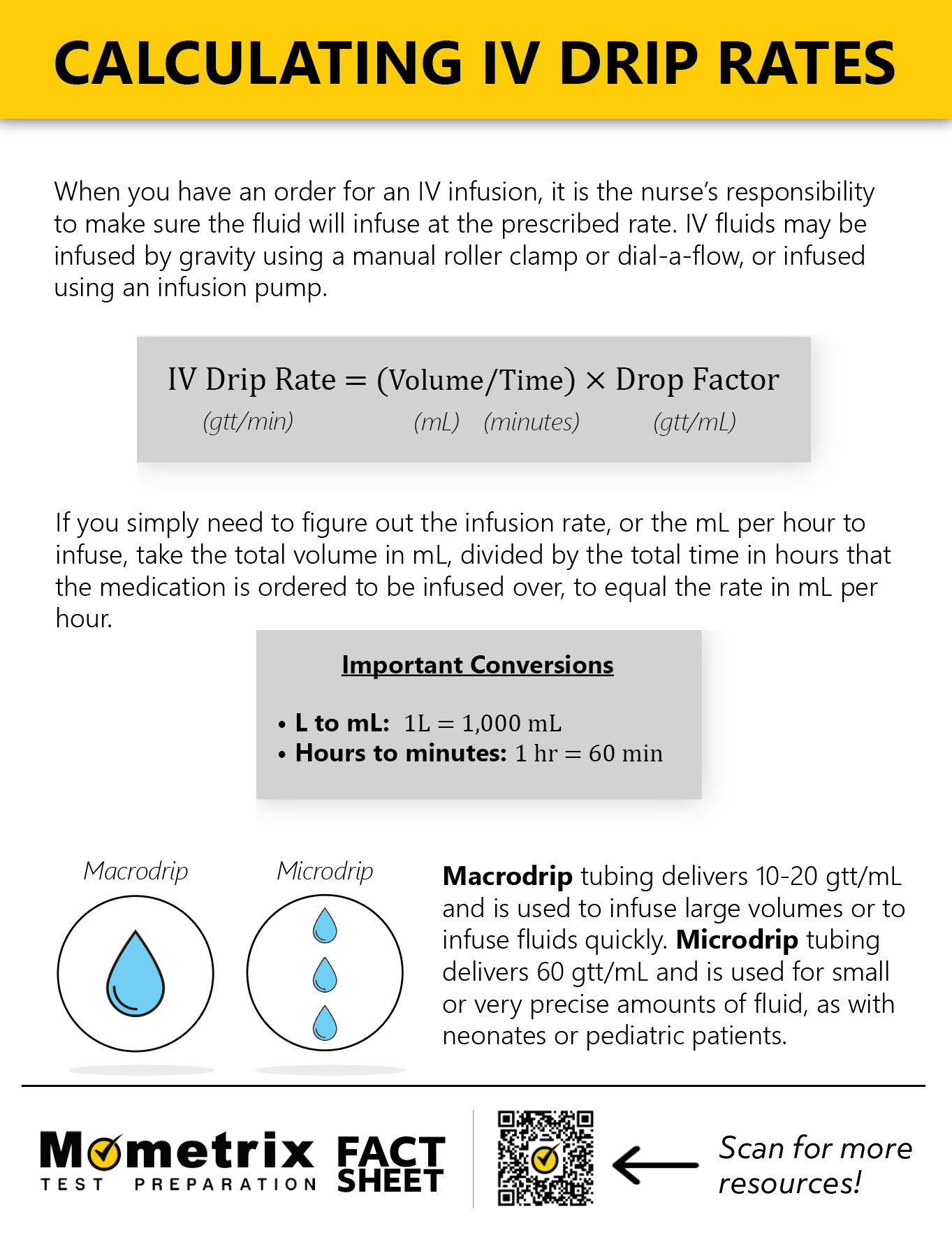
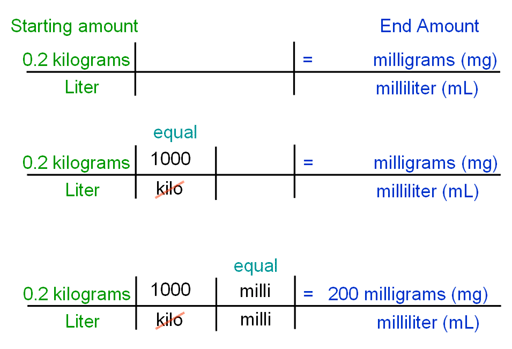
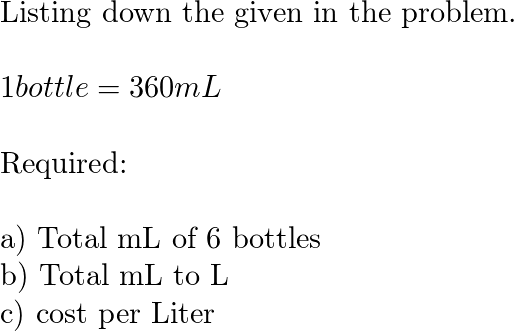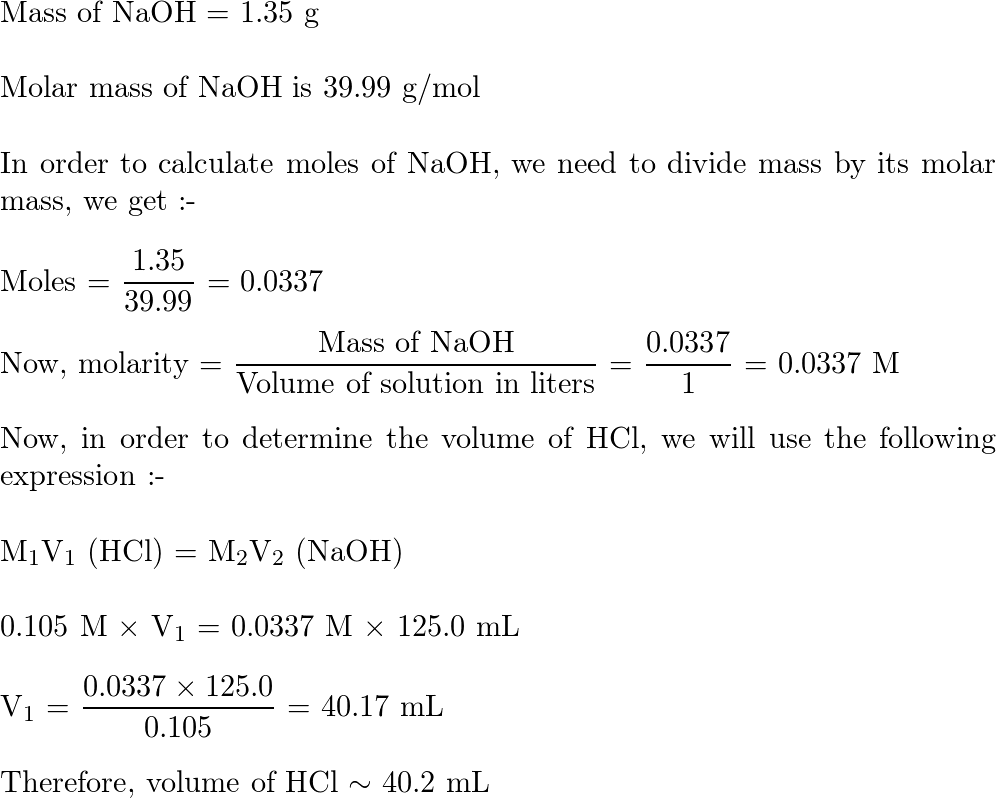PPT - MOLARITY A measurement of the concentration of a solution PowerPoint Presentation - ID:5814201

Shows Mean difference of TSH concentration in milli-international units... | Download Scientific Diagram

SOLVED: Convert 2.554 mg/mL into g/L Express your answer in grams per liter to four significant figures Azd g/L Submit Request Answer Part H Convert 2.554 mg/mL into pg/mL Express your answer

MOLARITY A measurement of the concentration of a solution Molarity (M) is equal to the moles of solute (n) per liter of solution M = mol / L Calculate. - ppt download
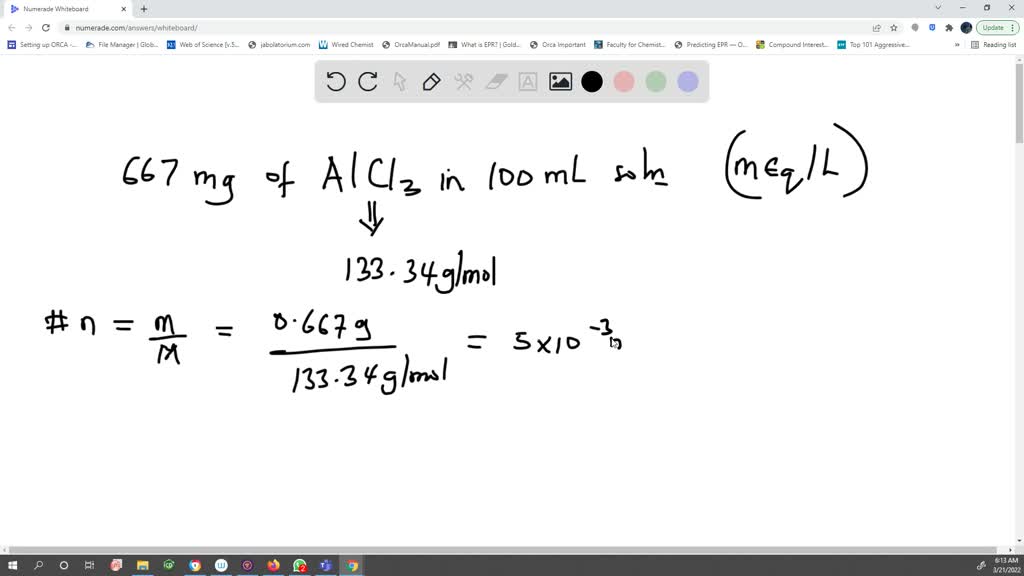
SOLVED: How many milliequivalents (mEq) per liter (mEq/L) are in 667 mg AlCl3 in 100 mL solution? 300 mEq/L 275 mEq/L 1650 mEq/L 0.275 mEq/L

PPT - mg/L - milligrams per litre (10-3 g per litre) = mg per 1,000,000mg THUS = ppm PowerPoint Presentation - ID:4270418








![How many ml in a liter? [Convert] - howmanyml.com How many ml in a liter? [Convert] - howmanyml.com](https://howmanyml.com/wp-content/uploads/2019/11/how-many-ml-equal-a-liter.jpg)