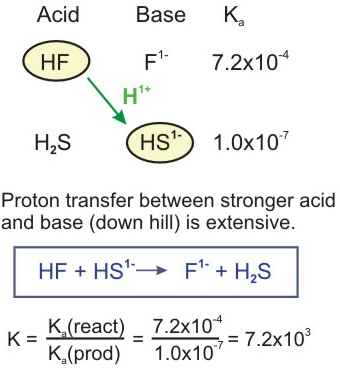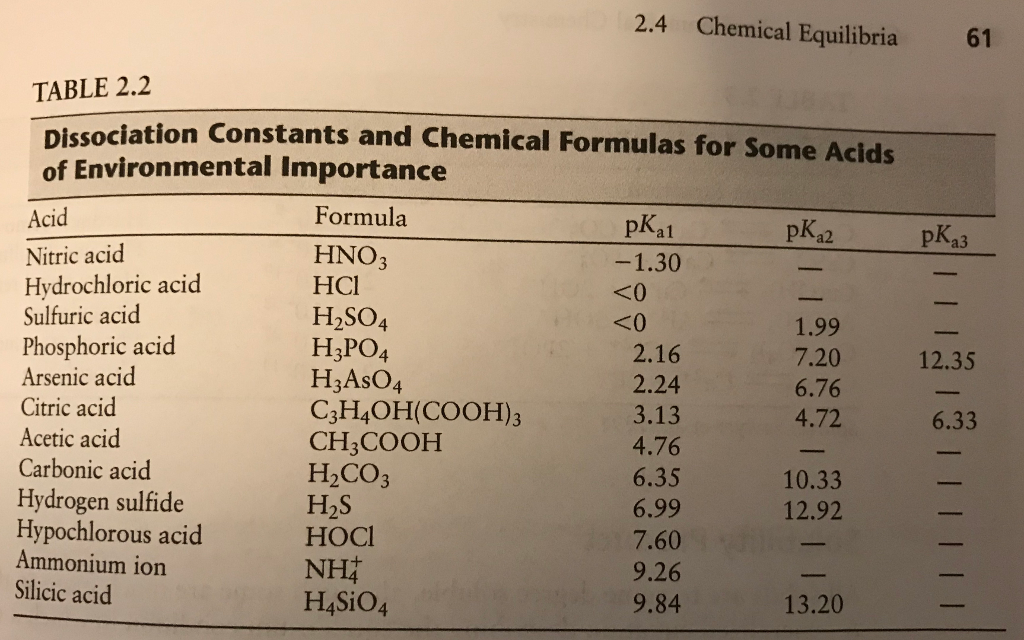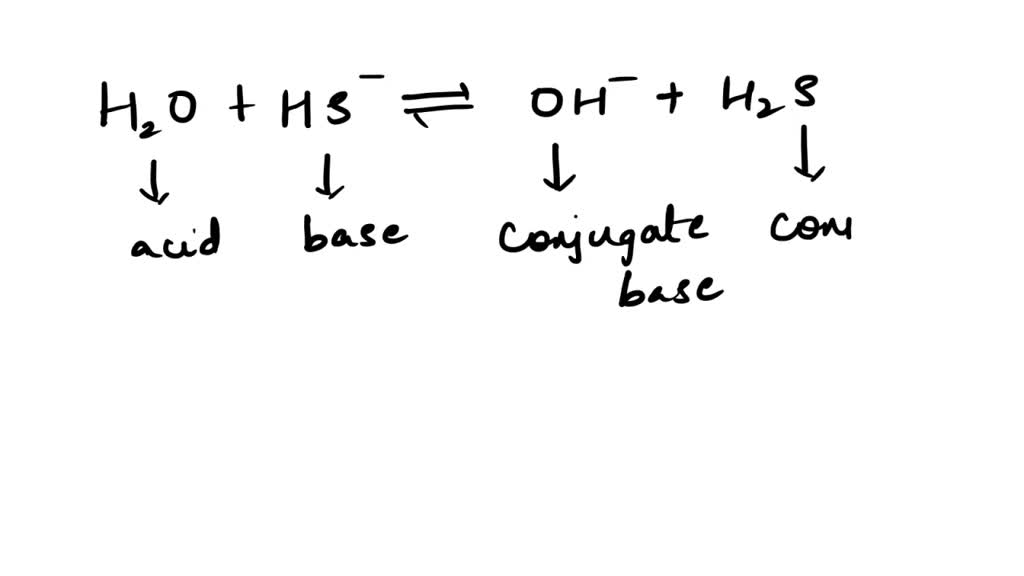
SOLVED: Be sure to answer all parts. In the following equation, identify the acids, bases, and conjugate pairs. H2O + HS− ⇌ OH− + H2S (a) What is the acid? H2O HS−

SOLVED: Write an equation that shows the reaction of hydrogen sulfide, HS– with hydroxide ion, OH–. Label the acid, the base, the conjugate acid, and the conjugate base.

Which of the species from the equilibrium below are conjugate acid-base pairs? NH3(aq) + H2S(aq) arrow HS-(aq) + NH4+(aq) | Homework.Study.com

Acid-base reaction with H2S liberation versus time. H2S detection via... | Download Scientific Diagram