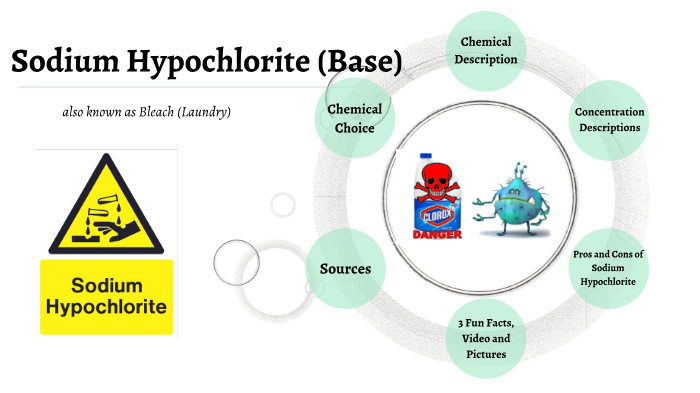
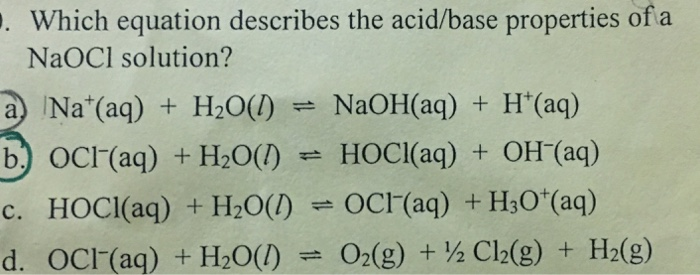Dissociation of chloramine-T and NaOCl. (A) In the presence of water... | Download Scientific Diagram

Dissociation of chloramine-T and NaOCl. (A) In the presence of water... | Download Scientific Diagram
Sodium Hypochlorite Pentahydrate Crystals (NaOCl·5H2O): A Convenient and Environmentally Benign Oxidant for Organic Synthesis

Sodium Hypochlorite Pentahydrate Crystals (NaOCl·5H2O): A Convenient and Environmentally Benign Oxidant for Organic Synthesis | Organic Process Research & Development

SOLVED: Match the following salts with the acid-base neutralization reactants that would produce them: A. NaClO4 = HCl + NaOH B. NaOCl = HCl + NaOH C. Ca(OCl)2 = HCl + Ca(OH)2