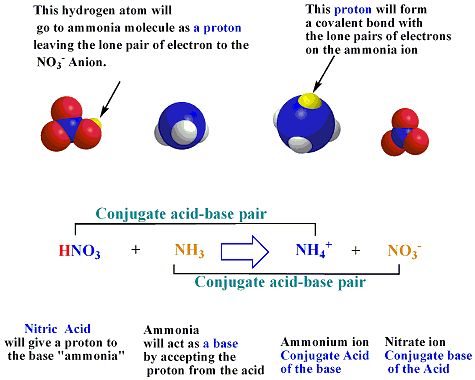
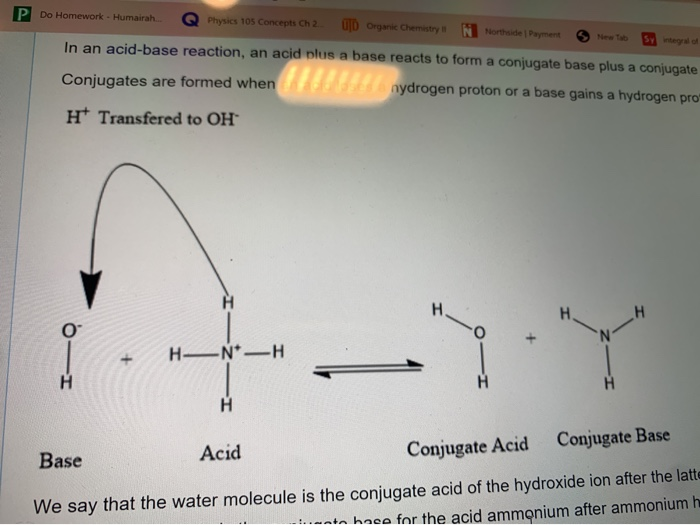
SOLVED: In the reaction NH3 (aq) + H2O (I) <–> NH4+ (aq) + OH- (aq), which is the conjugate acid-base pair? OH- NH3 NHA+, OH- NH3, H2O NH4+, NH3

Identify the conjugate acid-base pairs in this equilibrium. NH3(aq) + H2S(aq) arrow HS-(aq) + NH4+(aq) | Homework.Study.com
Why is NH4+ a weak acid? I have learned that the base and it's conjugate acid is opposite in strength, so why is NH4+ a weak acid when NH3 is also a