Photographers Mate Third Class (PH3) April Hatton, a photojournalist with Combat Camera Group, Atlantic, sharpens her skills on the .38 caliber pistol. Hatton has an expert ribbon on both the pistol and

Which of the following statements does not support the fact that NH3 is a stronger Lewis base than PH3 ?

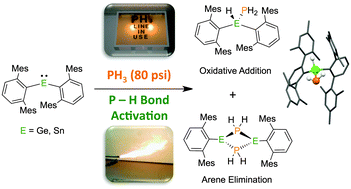
Taming PH3: State of the Art and Future Directions in Synthesis | Journal of the American Chemical Society

In which of the following arrangements, the sequence is not strictly according to the property written against it?

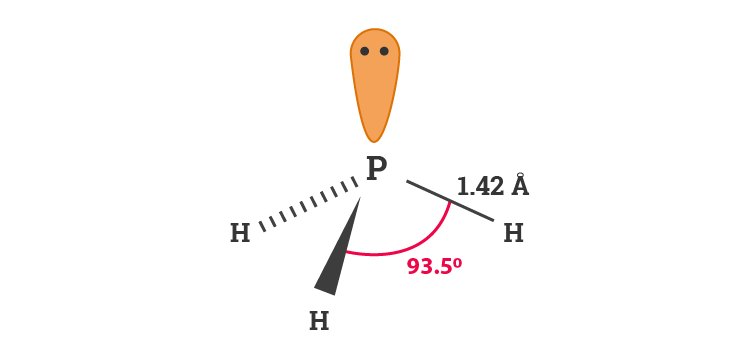
Draw the Lewis structure for PH3. How many bonds and non-bonding pairs are around the central atom, and what is the shape of this molecule? | Homework.Study.com

Mutations in Asn 125 of MYB.Ph3 influence base preferences at position... | Download Scientific Diagram

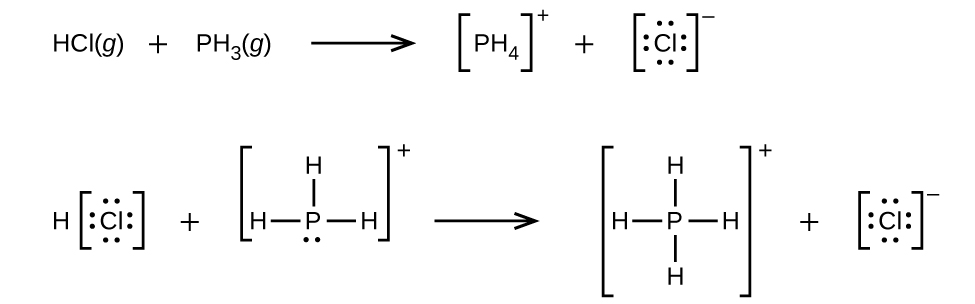
SOLVED: Identify the Lewis base in the following reaction: PH3(g) + H+(g) —> PH4+(g) Select one: a. None of these is a base b. PH4+ c. All of these are bases. d.

Draw the Lewis structure for PH3. How many bonds and non-bonding pairs are around the central atom, and what is the shape of this molecule? | Homework.Study.com
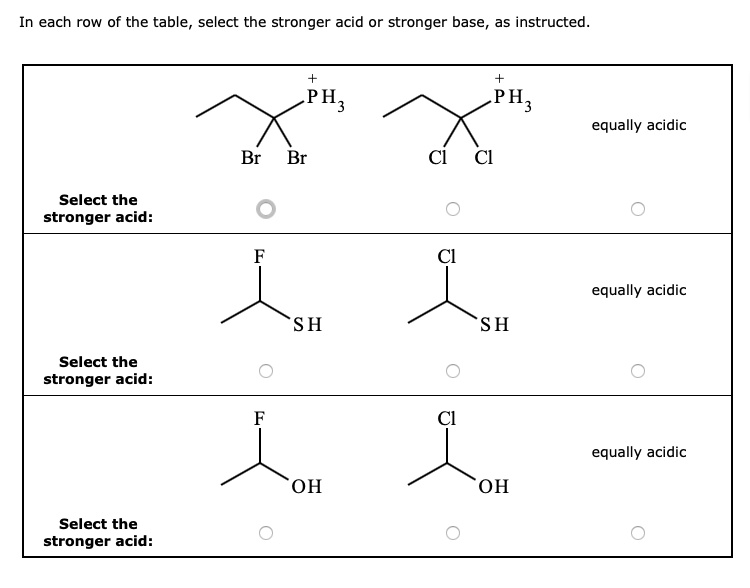
SOLVED: In each row of the table, select the stronger acid or stronger base as instructed. PH; PH3 equally acidic Br Br Select the stronger acid: equally acidic S H Select the